In the light of the world energy requirements for cleaner sources, the use of biogas has been raised as a promising alternative. Since biogas contains a higher hydrogen-to-carbon ratio, its combustion leads to reduced carbon dioxide emissions than fossil fuels. Owing to its nature, biogas requires a purification process (known as biogas upgrading) to allow its direct use in combustion engines, gas turbines, or fuel cells. The increased interest in biogas upgrading has generated the quest for technologies that can allow a more efficient process with a high-quality gas separation. Current technologies used for biogas upgrading are based either on sorption or separation processes; in this blog, we focus on sorption technologies.
Why does CO2 need to be removed?
.png?width=324&name=shutterstock_1690610593%20(2).png) Biogas is a product of the anaerobic digestion of organic matter by microorganisms. Raw biogas is a mixture of gases including methane, carbon dioxide, sulfur compounds, and water. Biogas upgrading aims to increase the methane content per unit of gas volume and is usually carried out in different steps. The separation starts by removing sulfur compounds and water, followed by a separation of methane and carbon dioxide. Since CO2 is the primary contaminant, its removal from biogas represents a critical step in economic terms. The required energy for the separation also impacts the overall efficiency of production. Therefore, the separation of CO2 from biogas is critical for the use of this fuel.
Biogas is a product of the anaerobic digestion of organic matter by microorganisms. Raw biogas is a mixture of gases including methane, carbon dioxide, sulfur compounds, and water. Biogas upgrading aims to increase the methane content per unit of gas volume and is usually carried out in different steps. The separation starts by removing sulfur compounds and water, followed by a separation of methane and carbon dioxide. Since CO2 is the primary contaminant, its removal from biogas represents a critical step in economic terms. The required energy for the separation also impacts the overall efficiency of production. Therefore, the separation of CO2 from biogas is critical for the use of this fuel.
The technologies for CO2 sorption include adsorption and absorption processes; such operations are based on physical (temperature and pressure) and chemical (acid-base neutralization reactions) processes. Today's commercial technologies for biogas upgrading are water scrubbing, amine absorption, organic solvent sorption, membrane separation, and solid sorbent technologies. The most used current technology is absorption using liquid media such as amines. However, one of the major drawbacks of liquid sorbents is the low CO2 sorption capacity and the high energy requirements for carrier regeneration.
Alternative CO2 capture technologies
The implementation of solid adsorbents for CO2 capture has gained more attention in recent years. This technology is a more attractive alternative in energy-saving since it does not require high pressure or low temperature compared to other methods. In solid adsorbents, the target gas molecules (CO2 for this system) adhere to the surface while other components pass through. Since the process is dependent on the surface area, porous materials have been used to increase the capture capacity. For this matter, several porous materials have been considered to be useful as sorbents such as zeolites, carbonaceous materials (activated carbon, charcoal, graphene, or carbon nanotubes), and Metal-Organic Frameworks (MOFs).
Comparing porous adsorbents

The picture shows a zeolite on the left and a MOF on the right. Zeolites are by far the most studied porous materials, and they have been implemented for several adsorption processes at an industrial scale. The high chemical and thermal stability of zeolites are key for gas separation applications. The adsorption in zeolites is influenced by many factors such as their structure and composition, purity, size and shape of the molecules, and polarity. Moreover, operating conditions like pressure and temperature directly impact the adsorption kinetics, showing an enhancement as both increase. The surface area of zeolites ranges from about 450 to 900 m2/g. An example of the zeolite implementation for CO2 adsorption is zeolite Li-X which achieved an adsorption capacity of 5.36 mmol/g at 1 bar and 25 °C.
Carbonaceous materials offer the advantages of easy accessibility, low cost, high thermal stability, and low sensitivity to moisture. Carbon materials offer higher surface areas compared to zeolites as high as 2000 m2/g. Despite their high porosity, carbon materials have moderate adsorption capacity, even lower than those shown by zeolites at low pressure, along with a limited selectivity.
More recently, MOFs, a new class of hybrid crystalline materials, have appeared as a great alternative to typical porous materials. These materials consist of metal nodes connected by organic ligands. Their highly ordered nature allows the construction of three-dimensional networks with uniform pore size and shape with pore diameters in the range of 30-200 nm. Their unique structure gives them robustness, high thermal and chemical stability, along with a super high porosity that can be as high as 7000 m2/g surpassing the highest porosity reported for zeolites and carbon materials.
.png?width=1920&name=Biogas%20upgrade%20scheme%20(1).png)
The most important advantage of MOFs compared to their counterparts is the ability to fine-tune their composition through a change of the metal center or the organic ligand. The potential of MOFs for biogas upgrading has been tested, showing promising results. For instance, MOF-74, MOF-505, and Cu3(BTC)2 are MOFs reported with extremely high CO2 capacities at low pressure (ca. 1 bar). The selectivity for gas separation in porous materials is related to several factors, including molecular sieving, thermodynamic equilibrium, and kinetics. Besides the high porosity of MOFs, their high structure tunability allows adjusting features like pore size, channel topology, and inner surface properties, enhancing the selectivity of the adsorbent material. Studies showed that the adsorption selectivity of 4 ~6 for CO2 over CH4 at a pressure range of 0.1 ~3 bars and a high CO2 adsorption capacity (6.6 mmol/g at 2.5 bars and 30 °C) could be achieved by the implementation of MOFs.
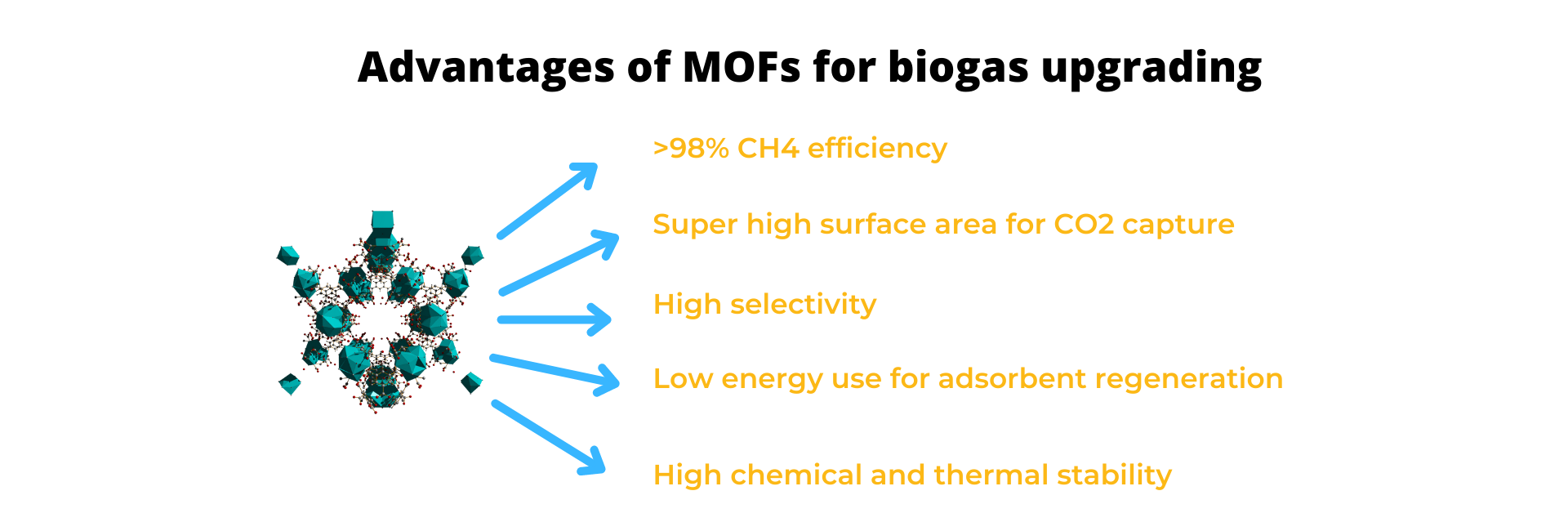
The implementation of solid adsorbents is a promising alternative for biogas upgrading showing high selectivity and capture capacity. Their use to obtain high-quality biomethane can help to reduce the energy consumption during the process, increasing the efficiency of the production process.
Are you interested in MOFs for gas capture? Please read more on carbon capture using MOFs in our previous blogs and case study.




.jpg)
