Nitroaromatic compounds (NACs) are a group of molecules of great importance as raw materials. NACs are used in the production of pesticides, pharmaceuticals, dyes, and explosives among other chemicals. Special interest has been put on the development of detection techniques for NACs since they represent a risk to human health and safety.
Current methods for the detection of nitroaromatic compounds have high sensitivity and selectivity (i.e. gas chromatography coupled to mass spectrometry and surface-enhanced Raman spectroscopy). However, they are of high cost and not portable for in situ detection. Therefore, the necessity of low cost and portable devices for NACs detection remains in the scope of research for real applications.
Challenges in the detection of nitroaromatic compounds
The chemical nature of NACs can be described as a benzene ring functionalized with different nitro-groups. These molecules show low to moderate vapor pressure and low chemical reactivity, making their detection in real-world applications challenging.
In recent years, Metal-Organic Frameworks (MOFs) have raised awareness as alternative materials for sensing technology. Their porous nature and the ability to fine-tune their chemical reactivity make them promising candidates for gas sensors. There are several applications for MOF-based sensors in gas samples including the detection of toxic and hazardous molecules (CO, NO, NO2, and SO2).
MOF-based sensors for nitroaromatic compound detection
 Luminescent MOFs (LMOFs) are a type of Metal-Organic Frameworks that incorporate a light-emitting component (ion, ligand, or a guest molecule). The change in the luminescence intensity of these MOFs is used as a signal for the detection of the target molecule. The interaction of the analyte with the MOF structure can increase or decrease the intensity of the luminescence. When the analyte increases the intensity, the sensor is a so-called “turn on” switch. When the reaction decreases the intensity a “turn off” sensor is created (this phenomenon is known as quenching).
Luminescent MOFs (LMOFs) are a type of Metal-Organic Frameworks that incorporate a light-emitting component (ion, ligand, or a guest molecule). The change in the luminescence intensity of these MOFs is used as a signal for the detection of the target molecule. The interaction of the analyte with the MOF structure can increase or decrease the intensity of the luminescence. When the analyte increases the intensity, the sensor is a so-called “turn on” switch. When the reaction decreases the intensity a “turn off” sensor is created (this phenomenon is known as quenching).
The porosity of the MOFs materials in one characteristic allowing high sensitivity. The capture of the target molecules inside the pores increases the changes of chemical interactions. Besides, this adsorption can function as a pre-concentration step, which can help to lower the limit of detection of the sensor.
Similarly, selectivity is affected by the pore size of the MOFs (molecular sieving) and the high internal surface area. Also, chemical reactivity can be easily manipulated (by changing the metal center and the organic linkers) to increase selectivity even among the nitroaromatic compounds themselves.
Quick reaction, application in humid conditions and multi-detection
In real applications, high stability is a paramount feature for sensors. One of the main drawbacks of luminescence sensors (electrochemical and polymer-based sensors) for explosives detection is hydrothermal stability. A recent report showed a MOF-based sensor for the detection of benzene and nitrobenzene that can be stable after several uses in high humidity conditions (at 265 °C). Furthermore, the Cd-MOF sensor presents a dual function; a fluorescence increase is obtained in the presence of benzene, while a quenching effect is observed in the presence of nitrobenzene vapor.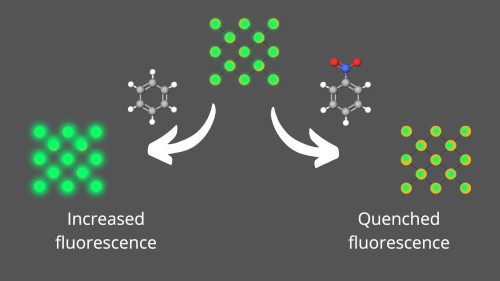
Another interesting feature of MOFs as sensing materials is that they have quick response times in the range from 30 to 180 seconds. The recovery of the sensor is possible under a simple thermal treatment at 150 °C for 60 s, showing 90 % signal recovery that makes them a promising candidate for real detection.
MOF sensors have been applied successfully for the detection of a wide variety of nitroaromatic compounds such as 1,3-dinitrobenzene (1,3-DNB), 1,4-dinitrobenzene (1,4-DNB), 2,4-dinitrotoluene (2,4-DNT), 2,6-dinitrotoluene (2,6-DNT), 2,4,6-trinitrotoluene (TNT), 2,4,6-trinitrophenol (TNP), nitrobenzene (NB), and 2-nitrotoluene (2-NT).
Conclusions
In summary, the properties of MOF can offer several advantages for the selective and sensitive detection of explosives in the gas phase.
- Chemical and structural tunability – Help to achieve good selectivity through molecular sieving effects or host-guest interactions.
- High internal surface – Concentrate the analytes to obtain high density that allows decreasing the detection limits to offer high sensitivity.
- Chemical and hydrothermal stability – Offer reliability and reuse of the sensor making them adequate for real applications.
MOF-based sensors are a promising alternative to expensive and time-consuming methods for the detection of explosive molecules. In contrast to other luminescent methods offer high chemical and thermal stability under high humidity conditions. The selectivity and sensitivity can be increased by fine-tuning of the pore size and chemical reactivity of the MOF structure. The implementation of Metal-Organic Frameworks as gas sensor materials result in low cost and portable devices that meet the reliability and reuse capabilities necessary for the detection of nitroaromatic compound used in explosive fabrication.





