Summer has been real short for us. Besides many projects with clients, we further increased our team. Read more about what we have been up to and what interesting publications and news we found around MOFs.
We welcome two new team members on board:

Anna Chomiak
As an application engineer, she will support our customers with her wide knowledge and experiences.

Oliver Maurer
He will support us with his background in production and laboratories in different chemical companies.
And then MOF2020web happened! An offline conference turned digital! It was awesome to gather with academics and advance our knowlegde again. Or as Omar Yaghi puts it:
“Although I feel that there is no substitute for meeting fellow scientists in person, the online MOF2020web succeeded in attracting many emerging scholars and newcomers to reticular chemistry (MOF/COF/ZIF fields) and they were engaged and inspired by the many interesting and stimulating contributions. Thus, it is definitely a worthwhile effort to have this online conference.”
Stay home, stay safe, but still enjoy autumn!
Startus Insights picked us for Top 5 materials startups!
They identified 44 startups that innovate the chemical industry with MOFs and picked novoMOF to be in their Top 5 showcase. It’s an honour to be recognised globally for the work that we do!
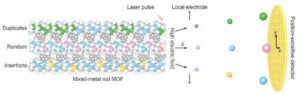
Furthermore, check out this article on sequencing multi-metal MOFs in order embed information in them:
Hidden atomic patterns discovered in mixed-metal MOFs
Multi-metallic materials could be encoded with instructions for synthesis or separation, that could form cascading processes.
From our blog
New articles of the last quarter

Energy storage: Improving batteries and supercapacitors
Improving batteries and supercapacitors in terms of service lifetime, energy density, and safety.

Electronic Devices: Application of Metal-Organic Frameworks
The miniaturization trend in electronic devices extends to the point of molecular-scale functionality in the nanometer scale.

Sensing nitroaromatic explosives in the gas phase
Nitroaromatic compounds (NACs) are a group of molecules of great importance as raw materials, that present a risk to human health and safety. Detection is of utmost importance
Interesting publications
We have categorised the publications and news in the following sections (click to skip to the section):
Zeolitic Imidazolate Framework-Derived Ordered Pt–Fe Intermetallic Electrocatalysts for High-Performance Zn-Air Batteries
Aiming to explore novel oxygen reduction reaction (ORR) catalysts with advanced electrochemical performance and economical and long-term stability is crucial to fuel cells or metal-air batteries. Herein, ordered Pt–Fe intermetallic with an ultralow Pt loading is facilely achieved via the thermal treatment of Pt nanocrystals anchored to the Fe, N-codoped surface-functioned carbon derived from zeolitic imidazolate frameworks. Benefiting from the ordered Pt–Fe intermetallic formed by the rationally applied calcination treatment, the optimized catalyst shows a larger mass activity and better long-term durability compared with Pt/C. Besides, when the optimized sample is applied in the configuration of a zinc-air battery system, it has a higher maximum power density, a better specific capacity as well as a longer discharge time, surpassing those of the Pt/C catalyst.
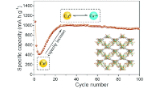
A green ligand-based copper–organic framework: a high-capacity lithium storage material and insight into its abnormal capacity-increase behavior
Metal–organic frameworks (MOFs) are believed to be potential lithium storage materials for their redox activity that can uptake electron and porosity that in turn can favor ion transport and storage. To boost their performance and practical use, in this work, a green and cheap ligand citrate is first used in constructing a Cu-based MOF anode. The as-prepared Cu-CIT exhibits excellent capacity, stability, and rate capability, with the capacities of 608.5 and 321.5 mA h g−1 at 1 and 2 A g−1 after 500 cycles, being the best one among Cu-based MOFs. Moreover, its abnormal capacity-increase behavior is also observed and elaborately investigated through ex situ electron paramagnetic resonance (EPR) and X-ray absorption fine structure (XAFS). These results reveal the gradual redox participation of metal centers during cycles which for the first time shed light on the progressive activated process of MOFs in LIB applications.
Comparison of MIL-101(Cr) metal-organic framework and 13X zeolite monoliths for CO2 capture
A comparative study was conducted to determine the pore properties and adsorptive performance of monoliths containing either the MIL-101(Cr) metal-organic framework or 13X zeolite for carbon dioxide (CO2) capture. Although there have been a great deal of previous work on CO2 adsorption onto zeolites and MOFs, there has been far fewer studies on structured adsorbents such as monoliths. The results indicate that MIL-101(Cr) monoliths have 1.3 times higher porosity than 13X zeolite monoliths. Increasing CO2 partial pressure in the gas mixture shortens breakthrough and equilibrium times and increases the breakthrough and equilibrium adsorption capacities of CO2. MIL-101(Cr) monoliths show better mass transfer of CO2 in the adsorbent bed with shorter breakthrough and equilibrium times of about 20% and 35%, respectively, than 13X zeolite monoliths. The adsorption capacity of CO2 on MIL-101(Cr) monoliths is higher by about 37% (based on weight in mmol/g) at breakthrough and slightly lower by about 7% at equilibrium when compared to 13X zeolite monoliths. MIL-101(Cr) monoliths were found to be 1.5 times more efficient for CO2 adsorption than 13X zeolite monoliths. The effects of regeneration temperature after CO2 adsorption on MIL-101(Cr) and 13X zeolite monoliths were studied and results showed an increase in CO2 adsorption capacity as the regeneration temperature was increased. In summary, the study showed MIL-101(Cr) monoliths have better CO2 adsorption properties than 13X zeolite monoliths.
Effect of framework rigidity in metal-organic frameworks for adsorptive separation of ethane/ethylene
Ethane-selective adsorbents having high adsorption capacities along with high selectivity toward C2H6 are required to develop a cost-effective adsorption process for the facile separation of ethane (C2H6) from the ethane and ethylene (C2H4) mixture. Understanding the roles of physicochemical properties of the C2H6-selective adsorbent in terms of its C2H6/C2H4 separation performance is especially essential for the design of novel advanced materials. Herein, we selected the known metal-organic framework structure, namely, the pillared three-dimensional framework DUT-8, which is synthesized by incorporation of 2,6-naphthalenedicarboxylic acid as a linker. We compared physicochemical properties of DUT-8 according to the framework flexibility and the type of metal ion. Even though the framework stability of DUT-8 is very low in the presence of water vapor, we further evaluated the C2H6/C2H4 separation performance of isomorphous DUT-8(M) adsorbents with different metal ions (M = Co, Ni, Cu, and Zn) by means of single-gas isotherm measurements, ideal adsorbed solution theory calculations, grand canonical Monte Carlo simulations, and density functional theory calculations. Separation results showed that preferential uptakes of C2H6 were observed from the C2H6/C2H4 mixture particularly over the rigid framework of DUT-8 (Cu and Ni) through the C–H‧‧‧π interaction close to the metal node. Thus, high selectivity of C2H6 was achieved over DUT-8(Cu and Ni) along with high uptakes of C2H6 (up to 11.2 mmol/g) at a temperature range of 283–303 K and pressure of 10 bar. The breakthrough separation results on DUT-8(Cu) showed that the binary mixture of C2H6/C2H4 (1 : 9 v/v) can be effectively separated at pressures of 1 and 5 bar with high productivity of high purity ethylene (20.8 L/kg at 1 bar and 45.0 L/kg at 5 bar). The facile regeneration possibility over DUT-8 is confirmed by the dynamic desorption studies, expecting continuous production of high purity ethylene.
Ultrafast Semi‐Solid Processing of Highly Durable ZIF‐8 Membranes for Propylene/Propane Separation
ZIF‐8 membranes have emerged as the most promising candidate for propylene/propane (C3H6/C3H8) separation through its precise molecular sieving characteristics. The poor reproducibility and durability, and high cost, thus far hinder the scalable synthesis and industrial application of ZIF‐8 membranes. Herein, we report a semi‐solid process featuring ultrafast and high‐yield synthesis, and outstanding scalability for reproducible fabrication of ZIF‐8 membranes. The membranes show excellent C3H6/C3H8 separation performance in a wide temperature and pressure range, and remarkable stability over 6 months. The ZIF‐8 membrane features dimethylacetamide entrapped ZIF‐8 crystals retaining the same diffusion characteristics but offering enhanced adsorptive selectivity for C3H6/C3H8. The ZIF‐8 membrane was prepared on a commercial flat‐sheet ceramic substrate. A prototypical plate‐and‐frame membrane module with an effective membrane area of about 300 cm2 was used for efficient C3H6/C3H8 separation.
Efficient Trapping of Trace Acetylene from Ethylene in an Ultramicroporous Metal–Organic Framework: Synergistic Effect of High‐Density Open Metal and Electronegative Sites
Acetylene (C2H2) removal from ethylene (C2H4) is a crucial step in the production of polymer‐grade C2H4 but remains a daunting challenge because of the similar physicochemical properties of C2H2 and C2H4. Currently energy‐intensive cryogenic distillation processes are used to separate the two gases industrially. A robust ultramicroporous metal–organic framework (MOF), Ni3(pzdc)2(7 Hade)2, is reported for efficient C2H2/C2H4 separation. The MOF comprises hydrogen‐bonded linked one‐dimensional (1D) chains, and features high‐density open metal sites (2.7 nm−3) and electronegative oxygen and nitrogen sites arranged on the pore surface as cooperative binding sites. Theoretical calculations, in situ powder X‐ray diffraction and Fourier‐transform infrared spectroscopy revealed a synergistic adsorption mechanism. The MOF possesses S‐shaped 1D pore channels that efficiently trap trace C2H2 at 0.01 bar with a high C2H2 uptake of 60.6 cm3 cm−3 and C2H2/C2H4 selectivity.
High Ammonia Uptake of a Metal‐Organic Framework Adsorbent in a Wide Pressure Range
Although numerous porous adsorbents have been investigated for NH 3 capture applications, these materials often exhibit insufficient NH 3 uptake, low NH 3 affinity at the ppm level, and poor chemical stability against wet NH 3 conditions. Herein, we report the NH 3 capture properties of M 2 (dobpdc) complexes (M = Mg 2+ , Mn 2+ , Co 2+ , Ni 2+ , and Zn 2+ ; dobpdc 4− = 4,4‐dioxidobiphenyl‐3,3‐dicarboxylate) that contain open metal sites. The NH 3 uptake of Mg 2 (dobpdc) at 298 K was 23.9 mmol g −1 at 1 bar and 8.25 mmol g −1 at 570 ppm, which are record high capacities at both pressures among existing porous adsorbents. The strength of the interactions between the Lewis acid open metal sites and NH 3 followed the trend of Ni > Mg > Co > Mn, which was determined by NH 3 ‐temperature programmed desorption curves. The structural stability of Mg 2 (dobpdc) upon exposure to wet NH 3 was superior to that of the other M 2 (dobpdc) and the frameworks tested. Our van der Waals‐corrected density functional theory calculations predicted that this may be associated with the higher stability of MgO 6 coordination compared to that of Mg(NH 3 ) 6 . Overall, these results demonstrate that Mg 2 (dobpdc) is a recyclable compound that exhibits significant NH 3 affinity and capacity, making it a promising candidate for real‐world NH 3 ‐capture applications.
An Ultramicroporous Metal–Organic Framework for High Sieving Separation of Propylene from Propane
Highly selective adsorptive separation of olefin/paraffin through porous materials can produce high purity olefins in a much more energy-efficient way than the traditional cryogenic distillation. Here we report an ultramicroporous cobalt gallate metal–organic framework (Co-gallate) for the highly selective sieving separation of propylene/propane at ambient conditions. This material possesses optimal pore structure for the exact confinement of propylene molecules while excluding the slightly large propane molecules, as clearly demonstrated in the neutron diffraction crystal structure of Co-gallate⊃0.38C3D6. Its high separation performance has been confirmed by the gas sorption isotherms and column breakthrough experiments to produce the high purity of propylene (97.7%) with a high dynamic separation productivity of 36.4 cm3 cm–3 under ambient conditions. The gas adsorption measurement, pore size distribution, and crystallographic and modeling studies comprehensively support the high sieving C3H6/C3H8 separation in this MOF material. It is stable under different environments, providing its potential for the industrial propylene purification.
Computational Selection of High-Performing Covalent Organic Frameworks for Adsorption and Membrane-Based CO2/H2 Separation
Covalent organic frameworks (COFs) have high potential in gas separation technologies because of their porous structures, large surface areas, and good stabilities. The number of synthesized COFs already reached several hundreds, but only a handful of materials were tested as adsorbents and/or membranes. We used a high-throughput computational screening approach to uncover adsorption-based and membrane-based CO2/H2 separation potentials of 288 COFs, representing the highest number of experimentally synthesized COFs studied to date for precombustion CO2 capture. Grand canonical Monte Carlo (GCMC) simulations were performed to assess CO2/H2 mixture separation performances of COFs for five different cyclic adsorption processes: pressure swing adsorption, vacuum swing adsorption, temperature swing adsorption (TSA), pressure−temperature swing adsorption (PTSA), and vacuum−temperature swing adsorption (VTSA). The results showed that many COFs outperform traditional zeolites in terms of CO2 selectivities and working capacities and PTSA is the best process leading to the highest adsorbent performance scores. Combining GCMC and molecular dynamics (MD) simulations, CO2 and H2 permeabilities and selectivities of COF membranes were calculated. The majority of COF membranes surpass Robeson’s upper bound because of their higher H2 permeabilities compared to polymers, indicating that the usage of COFs has enormous potential to replace current materials in membrane-based H2/CO2 separation processes. Performance analysis based on the structural properties showed that COFs with narrow pores [the largest cavity diameter (LCD) < 15 Å] and low porosities (ϕ < 0.75) are the top adsorbents for selective separation of CO2 from H2, whereas materials with large pores (LCD > 20 Å) and high porosities (ϕ > 0.85) are generally the best COF membranes for selective separation of H2 from CO2. These results will help to speed up the engineering of new COFs with desired structural properties to achieve high-performance CO2/H2 separations.
Thermochemical Analysis of Ammonia Gas Sorption by Struvite from Livestock Wastes and Comparison with Biochar and Metal–Organic Framework Sorbents
Struvite-bearing solids from swine (S) and dairy (D) wastewater, heat-treated to 150–300 °C, were evaluated as ammonia gas (NH3(g)) sorbents and compared to biochar (BC) and a metal–organic framework (MOF). Simultaneous thermal analysis-pulse thermal analysis-Fourier-transform infrared spectroscopy (STA-PTA-FTIR) was used to determine sorption capacity, reversibility, thermodynamics, and kinetics. For wastewater-derived sorbents, S solids heated to 150 °C (S-150) had the highest NH3(g) sorption capacity (47.2–49.9 mg g–1), comparable to BC (50.8 mg g–1). Enthalpies increased with sorption capacity, and the energy released per mole sorbed NH3(g) indicated stronger bonds formed with S sorbents than BC. After desorption, S-150 retained more NH3(g) (48–51%) than BC (39%). The MOF had the highest sorption capacity (289.7 mg g–1) and irreversibly bound NH3(g) (81%) but similar sorption activation energy (Ea) as S-150. The rates (k) of NH3(g) sorption and desorption were fastest for S-150. Overall, S-150 sorbents performed similarly to BC but were less effective than MOF for NH3(g) sequestration. However, advantages of S-150 for NH3(g) mitigation include wastewater valorization, minimal synthesis, low heat treatment, and potential use in agricultural applications. Evaluation of struvite-based wastewater-derived sorbents, comparison with commonly used sorbents, and the implementation of thermochemical analysis for this purpose are all novel aspects of this study.
Bioinspired Engineering of a Bacterium‐Like Metal–Organic Framework for Cancer Immunotherapy
Bacteria‐mediated tumor therapy (BMTT) has been known for decades; however, its clinical use is inhibited by its association with infections. To address this issue, a spiky, bacterium‐like metal–organic framework (MOF), which can replicate the functional responses of BMTT without its adverse side‐effects, is proposed. MOFs are synthesized in a solvothermal reaction of aluminum sulfate, ruthenium chloride hydrate, and 2‐aminoterephthalic acid; they have a spherical morphology or many nanospikes on their surfaces, depending on the reaction temperature. Both spherical and spiky MOFs can function as photothermal agents, converting absorbed optical energy into local heat. Owing to their higher surface area of interaction, spiky MOFs are more easily phagocytosed by macrophages than are spherical MOFs, strengthening their immune responses. Moreover, when injected intratumorally, spiky MOFs reside significantly longer than spherical ones, enabling their use in repeated photothermal treatments. The combination of in situ vaccination with intratumorally injected bacterium‐like MOFs under exposure to an near‐infrared laser and the immune checkpoint blockade of systemically administered αPD‐1 is evaluated in tumor‐bearing mice. The results indicate that the checkpoint blockade acts synergistically with in situ vaccination to provide diverse antitumor functions of BMTT, destroying a primary tumor and suppressing tumor recurrence and metastasis.
De novo synthesis and particle size control of iron metal organic framework for diclofenac drug delivery
Metal organic frameworks (MOFs) possess lots of potential but what limits its applicability in the industrial setting is the sustainability of its preparation. This work features room temperature synthesis of MIL-100(Fe) without using toxic organic solvents and strong acids. Using varying ratio of trimesic acid (BTC) to diclofenac (DCF) as ligand during synthesis, DCF was directly incorporated within the MIL-100(Fe) framework. The products synthesized showed similar PXRD patterns with the calculated value for MIL-100(Fe). The de novo synthesized products (D1-D5) were compared to conventional DCF loading in MIL-100(Fe) (DCF@MIL) wherein they show comparable characteristics. SEM images also confirmed that the octahedral morphology of MIL-100(Fe) was preserved even when DCF substituted some BTC as ligand. DCF release in simulated body fluids were determined and showed sustained release over a 24-h period at pH 7.4. On the other hand, negligible release was observed at pH 1.2 which is favorable as DCF can be protected from enzymatic degradation in the stomach, thus more DCF can be delivered to the systemic circulation. Among the synthesized bio-MOFs, D2 showed promising results with 20% loading capacity and 94% entrapment efficiency. Controlled particle size (100–400 nm) with varied reaction time was achieved with DCF acting as modulator. Results showed that longer reaction time yielded larger particle size, higher porosity, and enhanced drug release. This study demonstrates the versatility of DCF wherein aside from being the active drug, it may also act as modulator in MOF synthesis for particle size control.
Porous materials as carriers of gasotransmitters towards gas biology and therapeutic applications
The discovery of NO, CO, and H2S as gasotransmitters and their beneficial role in multiple physiological functions opened an era of research devoted to exogenously delivering them as therapeutic agents. However, the gaseous nature of these molecules demands new forms of administration that enable one to control the location, dosage and timing of their delivery. Porous materials are among the most suitable scaffolds to store, deliver and release gasotransmitters due to their high surface area, tunable composition and reactivity. This review highlights the strategies employed to load and release gasotransmitters from different kinds of porous materials, including zeolites, mesoporous silica, metal–organic frameworks and protein assemblies.
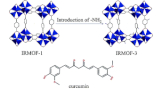
Amino-functionalized Zn metal organic frameworks as antitumor drug curcumin carriers
The tunability of their structure and function is why metal organic frameworks (MOFs) are widely used in drug carrier research. The modification of organic ligands can achieve the regulation of the functions and properties of MOFs. In our research, the amino-functionalized metal organic framework of Zn with 2-aminoterephthalate, IRMOF-3, was studied as an antitumor drug curcumin carrier. The nanoparticles can efficiently encapsulate the anticancer drug curcumin. IRMOFs and drug-loaded IRMOFs were characterized using different techniques. Furthermore, a simulated release test was carried out on the nanomedicine system in vitro. In addition, the drug loading and release activity has been compared with that of the isostructural amino-free MOF (IRMOF-1). The drug load capacity of IRMOF-3 (55.36%) is found to be significantly higher than that of IRMOF-1 (49.30%). And the cumulative release rate of curcumin from IRMOF-3 was slower than that from IRMOF-1. That is to say, IRMOF-3 has a more pronounced slow release behavior than IRMOF-1. The superior performance of IRMOF-3 has been attributed to the affinity between the side chain amino group and curcumin. Amino groups increase the affinity of the MOFs with curcumin, which can increase the drug loading capacity and slow the drug release rate. Also, the low cytotoxicity and good biocompatibility of the two materials were certified using an MTT assay. Thus, the addition of groups will have a certain effect on the drug loading performance of MOFs. We propose to synthesize different group-functionalized metal organic frameworks to obtain excellent drug carrier materials.
Designing inhalable metal organic frameworks for pulmonary tuberculosis treatment and theragnostics via spray drying
Inhalable metal organic framework (MOF) aerosols have been developed via spray drying as a therapy for multi-drug resistant (MDR) tuberculosis (TB). The CuPOA2 (pyrazinoate acid) MOFs can be tailored to exhibit a respirable mass median aerodynamic diameter (MMAD) of 2.6 μm. This method is repeated to manufacture Gd0.1Cu0.9(POA)2 MOFs for inhalable theragnostics.
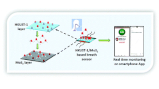
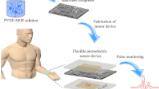
MOF based flexible, low-cost chemiresistive device as a respiration sensor for sleep apnea diagnosis
The monitoring of respiratory disorders requires breath sensors that are fast, robust, and convenient to use and can function under real time conditions. A MOF based flexible sensor is reported for the first time for breath sensing applications. The properties of a highly porous HKUST-1 MOF and a conducting MoS2 material have been combined to fabricate an electronic sensor on a flexible paper support for studying sleep apnea problems. Extensive breath sensing experiments have been performed and interestingly the fabricated sensor is efficient in detecting various kinds of breaths such as deep, fast, slow and hydrated breath. The MOF breath sensor shows a fast response time of just ∼0.38 s and excellent stability with no decline in its performance even after a month. A plausible mechanism has been proposed and a smartphone based prototype has been prepared to demonstrate the real time applications of the hybrid device. This work demonstrates great potential for the application of MOFs in healthcare with a special focus on breath sensing and sleep apnea diagnosis.
Toxic gas
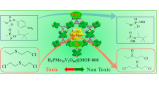
Heterometallic Titanium-Organic Frameworks as Dual-Metal Catalysts for Synergistic Non-buffered Hydrolysis of Nerve Agent Simulants
Mixed-metal or heterometallic metal-organic frameworks (MOFs) are gaining importance as a route to produce materials with increasing chemical and functional complexities. We report a family of heterometallic titanium frameworks, MUV-101(M), and use them to exemplify the advantages of controlling metal distribution across the framework in heterogeneous catalysis by exploring their activity toward the degradation of a nerve agent simulant of Sarin gas. MUV-101(Fe) is the only pristine MOF capable of catalytic degradation of diisopropyl-fluorophosphate (DIFP) in non-buffered aqueous media. This activity cannot be explained only by the association of two metals, but to their synergistic cooperation, to create a whole that is more efficient than the simple sum of its parts. Our simulations suggest a dual-metal mechanism reminiscent of bimetallic enzymes, where the combination of Ti(IV) Lewis acid and Fe(III)–OH Brönsted base sites leads to a lower energy barrier for more efficient degradation of DIFP in absence of a base.
Combining Two into One: A Dual-Function H5PV2Mo10O40@MOF-808 Composite as a Versatile Decontaminant for Sulfur Mustard and Soman
Due to the unpredictable nature of a battlefield environment, in the simultaneous degradation of sulfur mustard and nerve agents it is preferable to use just one decontaminant. Herein, the new composite HPVMo@MOF-808 (HPVMo = H5PV2Mo10O40) was deliberately synthesized via a simple impregnation method and thoroughly characterized. The results showed that the decontamination rate of the composites (30–40 mg) with optimal HPVMo loadings for HD (4 μL) and GD (4 μL) under ambient conditions was 97.2% (within 120 min) and 90.8% (within 30 min), respectively. Due to the combinational/synergistic effect of MOF-808 and encapsulated homogeneously dispersed HPVMo, the composite can very efficiently oxidize HD to nontoxic products in a single system, while retaining the inherent excellence of MOF-808 in hydrolytically degrading GD. The decontamination process was found to follow first-order reaction kinetics, and the rate constant and half-life of the composite for HD and GD were 0.0231 min–1, 30.13 min and 0.0795 min–1, 8.72 min, respectively. In addition, experimental results in guinea pigs and Kunming mice used as animal models showed that the composite provided effective skin protection against HD and GD, showing great potential for application in skin decontamination and protection.
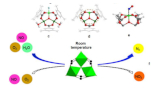
Room Temperature Reduction of Nitrogen Oxide to Nitrogen on MetalOrganic Frameworks
Air pollution is an epochal concern, particularly in urban areas, and is linked tocombustion processes. The emission of nitrogen oxides (NOx) constitutes acriticalenvironmental problem, and it can affect severely human health. At ambient temperature and pressure NOx decomposition is thermodynamically favoured; however, this process is kinetically inhibited, owing to a high activation energy. To date, no reported catalysts have had the required properties to lower the activation energy of this process without the help of co-reacting agents and high temperatures. Here, we show that NO conversion to molecular nitrogen can be achieved at room temperature in the presence of O2 and H2O vapour, and in the absence of any further reducing agent, using iron-based Metal-Organic Frameworks(MOFs). Further, we demonstrate that MOFs work similarly to enzymes, but are stable in environments unfriendly to living matter. These findings open large perspectives on the solution of stringent problems in chemistry, such as the removal of pollutants or the activation of highly stable molecules.
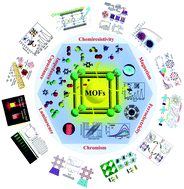
Functional metal–organic frameworks as effective sensors of gases and volatile compounds
Developing efficient sensor materials with superior performance for selective, fast and sensitive detection of gases and volatile organic compounds (VOCs) is essential for human health and environmental protection, through monitoring indoor and outdoor air pollutions, managing industrial processes, controlling food quality and assisting early diagnosis of diseases. Metal–organic frameworks (MOFs) are a unique type of crystalline and porous solid material constructed from metal nodes (metal ions or clusters) and functional organic ligands. They have been investigated extensively for possible use as high performance sensors for the detection of many different gases and VOCs in recent years, due to their large surface area, tunable pore size, functionalizable sites and intriguing properties, such as electrical conductivity, magnetism, ferroelectricity, luminescence and chromism. The high porosity of MOFs allows them to interact strongly with various analytes, including gases and VOCs, thus resulting in easily measurable responses to different physicochemical parameters. Although much of the recent work on MOF-based luminescent sensors have been summarized in several excellent reviews (up to 2018), a comprehensive overview of these materials for sensing gases and VOCs based on chemiresistive, magnetic, ferroelectric, and colorimertic mechanisms is missing. In this review, we highlight the most recent progress in developing MOF sensing and switching materials with an emphasis on sensing mechanisms based on electricity, magnetism, ferroelectricity and chromism. We provide a comprehensive analysis on the MOF–analyte interactions in these processes, which play a key role in the sensing performance of the MOF-based sensors and switches. We discuss in detail possible applications of MOF-based sensing and switching materials in detecting oxygen, water vapor, toxic industrial gases (such as hydrogen sulfide, ammonia, sulfur dioxide, nitrous oxide, carbon oxides and carbon disulfide) and VOCs (such as aromatic and aliphatic hydrocarbons, ketones, alcohols, aldehydes, chlorinated hydrocarbons and N,N′-dimethylformamide). Overall, this review serves as a timely source of information and provides insight for the future development of advanced MOF materials as next-generation gas and VOC sensors.
SERS‐Active MIL‐100(Fe) Sensory Array for Ultrasensitive and Multiplex Detection of VOCs
The application of metal–organic frameworks (MOFs) as SERS‐active platforms in multiplex volatile organic compounds (VOCs) detection is still unexplored. Herein, we demonstrate that MIL‐100 (Fe) serves as an ideal SERS substrate for the detection of VOCs. The limit of detection (LOD) of MIL‐100(Fe) for toluene sensing can reach 2.5 ppm, and can be even further decreased to 0.48 ppb level when “hot spots” in between Au nanoparticles are employed onto MIL‐100 (Fe) substrate, resulting in an enhancement factor of 1010. Additionally, we show that MIL‐100(Fe) substrate has a unique “sensor array” property allowing multiplex VOCs detection, with great modifiability and expandability by doping with foreign metal elements. Finally, the MIL‐100(Fe) platform is utilized to simultaneously detect the different gaseous indicators of lung cancer with ppm detection limit, demonstrating its high potential for early diagnosis of lung cancer in vivo.
Self-Powered Wearable Piezoelectric Sensors Based on Polymer Nanofiber–Metal–Organic Framework Nanoparticle Composites for Arterial Pulse Monitoring
High-performance wearable electronic devices with the capability of converting mechanical force into electrical energy have been gaining increasing attention for biomedical monitoring applications. We present a novel wearable piezoelectric sensor based on a poly(vinylidene fluoride) (PVDF) nanofibrous membrane containing microporous zirconium-based metal–organic frameworks (MOFs) for arterial pulse monitoring. It is shown that the incorporation of 5 wt % of MOF greatly enhances the piezoelectric constant of the polymer fibrous mat by 3.4-fold without significant loss in its flexibility. The nanofibrous composite exhibits a peak-to-peak voltage of 600 mV under an applied force of 5 N, which is superior to many flexible pressure sensors. It is demonstrated that the enhanced piezoelectric performance of the nanofibrous composite is not only attributed to the increased degree of crystallinity and polar β phase content (75%) but also to the surface chemistry and topography of the nanofibers. Evaluations of the piezoelectric output of the sensor attached to the radial artery at normal body conditions reveal significantly better output voltage (568 ± 76 mV) and sensitivity (0.118 V/N) than nanofibrous PVDF devices for wrist pulse monitoring. The results of this work pave a new way to develop flexible piezoelectric nanofibrous sensors based on MOFs for environmentally sustainable energy generation and wearable healthcare monitoring systems.
Megamerger of MOFs and g-C3N4 for energy and environment applications: upgrading the framework stability and performance
Metal–organic frameworks (MOFs) possess large surface area, adjustable pore size and synthetic adaptability which make them promising candidates for diverse applications. However, the low stability and selectivity of pure MOFs are not the optimal configuration for practical applications. Studies have confirmed that the controllable integration of MOFs with graphitic carbon nitride (g-C3N4) not only improves the stability and conductivity of pure MOFs, but also unexpectedly yields new functionalities. These advantages allow the synthesized g-C3N4/MOF-based materials to be applicable to fields other than pure MOFs and have attracted widespread attention in recent years. Herein, the review aims to specifically group together the important reports focused on g-C3N4/MOF-based materials till now. The synthetic methods and applications of composite materials are studied and classified. Furthermore, the review also highlights the functionalities produced by the synergy of two interesting materials, and finally paints a picture of the future.
Metal–Organic Framework-Intercalated Graphene Oxide Membranes for Highly Efficient Oil/Water Separation
Graphene oxide (GO) has drawn extensive research interest in the fabrication of high-performance membranes. However, the original narrow interlayer distance of the GO membrane restrains the permeation flux. Herein, a metal–organic framework (MOF)@GO nanocomposite was synthesized by growing UiO-66-NH2 (N-UiO-66), a water-stable MOF, on the GO nanosheet followed by modification with poly(acrylic acid) (PAA). MOF-intercalated GO membranes were fabricated by intercalating N-UiO-66@GO-PAA nanocomposites into adjacent GO nanosheets through the vacuum-assisted self-assembly process. Because of low membrane thickness and enlarged interlayer distance, the membranes showed significantly increased water permeance (5067 L m–2 h–1 bar–1 under optimum conditions) as well as high rejections for oil/water emulsion (>99.9%). Moreover, due to the hydrophilic chemical structures and typical micronano-topological structures, the MOF-intercalated GO membranes exhibited underwater superoleophobicity and antifouling property with a flux recovery ratio (FRR) above 80% after the three-cycle oil/water emulsion filtration test. Owing to the existence of multiple interactions, including electrostatic interactions and covalent and hydrogen bonds, the membranes exhibited satisfying operational and structural stability.
Filtering out toxic chromium from water
Hexavalent chromium continues to contaminate water sources around the world, with one US company fined just this February for having put employees at risk. Hexavalent chromium is considered to be extremely toxic, especially when inhaled or ingested, and its use is regulated in Europe and in many countries around the world. It is thought to be genotoxic, leading to DNA damage and the formation of cancerous tumors.
Now, chemists at EPFL are developing energy efficient processes for removing contaminants, this time hexavalent chromium, from water.













.jpg)
